Neonatal Biomagnetometer
A new investigational tool for neurological impairments of pre-term and full-term infants.babySQUID® (pdf) measures and maps brain activity non-invasively at the bedside.
CLINICAL ADVANTAGES of babySQUID®
- Map the sites and dynamics of sensory functions
- Map seizure and inter-ictal activity for epilepsy monitoring
- Assay stages of nervous system development
- Monitor recovery from trauma
- Detect effects of earlier hypoxic and intracranial injury
More newborns survive even with neurological disabilities.
CONDITION |
INCIDENCE |
| perinatal asphyxia | between 2 – 47 per1000 |
| hypoxemic-ischemic encephalopathy | between 3 – 8 per 1000 |
| moderate-to-severe cerebral palsy (post neonatal) | between 1 – 3 per 1000 |
| periventricular white matter injury | 240 per 1000 (for gestational age < 38 weeks) |
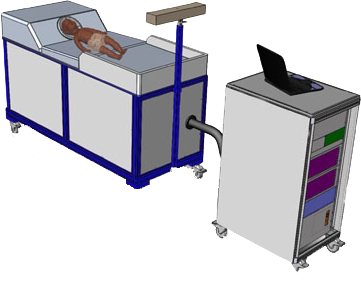
babySQUID® FEATURES
- Cradle measurement area fits infant’s head
- Close-spaced sensor array for superior spatial resolution
- Completely non-invasive
- Replaces EEG – no need to glue electrodes to infant’s scalp
- Portable – wheels from hospital elevator to bedside
- Needs no magnetically shielded room
- No need to immobilize the head
- Includes position tracking device and software
Principles of Operation
Superconducting amplifiers (SQUIDs) are used to amplify magnetic signals detected by a large array of small detection coils. The detection apparatus is kept at cryogenic temperatures by a vacuum insulated vessel (dewar). Tristan developed fabrication methods allow the detection coils to be placed extremely close to the patient without loss of sensitivity or risk to the patient. Additional information on neuromagnetic instrumentation can be found at Chapt2.pdf. The babySQUID® takes advantage of the fact that the infant’s scalp and skull are thin. This make it possible to measure MEG signals at a distance of only about 5-6 mm from the brain surface. This shorter distance results in a significant increase in amplitude of MEG signals from the newborns, since the magnetic field is inversely proportional to the square of the distance. The shorter distance and the high density of detectors also results in higher spatial resolution.
Advantages of BabySQUID®
- Unprecedented spatial resolution and sensitivity
A dense array of closely spaced sensors is located just below (<1 cm) the outer surface of a headrest. The sensor noise is <10 fT/√Hz for the detection coils - BabySQUID® has an order of magnitude better sensitivity to neuronal sources than conventional whole-head MEG systems. Sensitive enough to measure spontaneous neuronal activity and evoked activity of the cortex of the newborns in real time without signal averaging. Spatial resolution four times greater than existing whole-head MEG sensors.
- In comparison, EEG signals are significantly distorted by skull defects (fontanels and sutures) unique to the human neonates. These skull defects can obscure the asymmetry of the signals, especially when the generator is deep, making it difficult to determine the location of epileptiform tissue when it can not be easily visualized by CT or MRI.
Complete System
- 24 bit data acquisition hardware, operating under MS Windows
- Software acquisition at >5 kSamples/sec
- Optical positioning/tracking system
- Optical one-click 3D imaging
- Output ports for triggering sensory stimuli
- Data exports to BESA and EMSE software packages
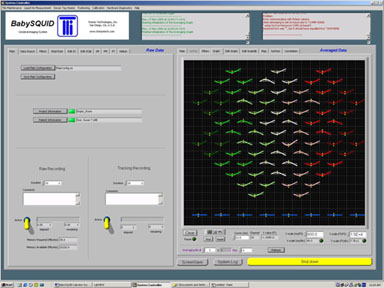
Simulated dipole data shown in sensor geometry layout
Power and Physical Requirements
- Power: 1 x 15 A circuit
- Patient Bed 1 m x 2 m, 500 kg
- Instrument Cart 19″ electronics rack, 150 kg
For additional information on babySQUID®, see babysquid_tech.pdf
Desciption of software interface see BabySQUID® Labview Sys Controls.pdf
For related adult human full head technologies, including clinical work on epilepsy, see:
babySQUID® is a registered trademark of Tristan Technologies, Inc. All rights reserved. This work funded by NIH under grant number: R44 NS38806 -03


